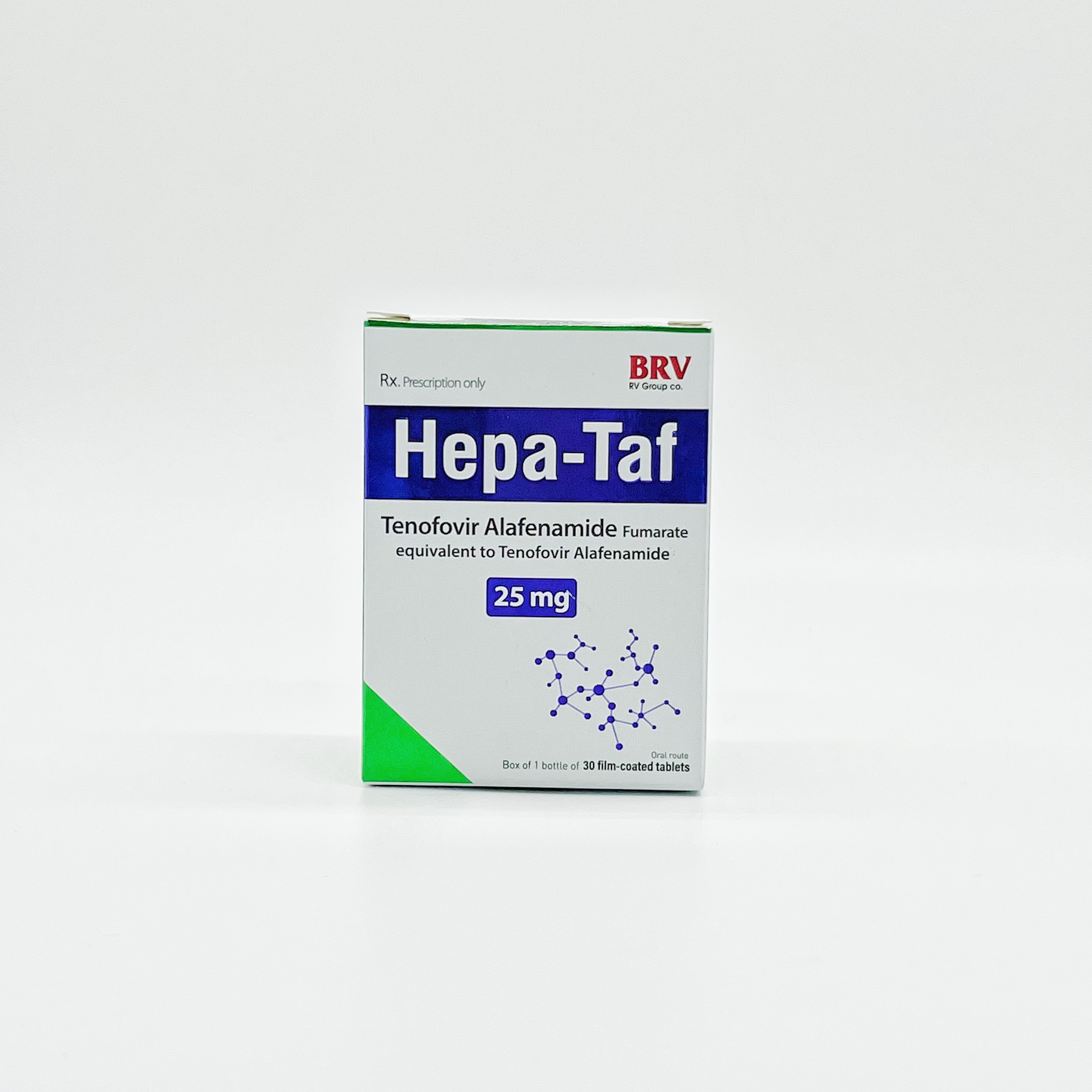
Hepa-taf
Description
Ingredients:
Tenofovir Alafenamide fumarate (equivalent to Tenofovir Alafenamide)
Excipients:
Lactose monohydrat, Cellulose microcrystals, Croscarmellose natri, Magnesi stearat, Opadry II white 85F18422.
Assign:
Tenofovir Alafenamide 25mg is indicated for the treatment of chronic hepatitis B in adults and teenager.
Expiry date:
24 months.
Packing:
Box of 1 bottle of 30 tablets.
Preservation:
Below 30°C.
Brand Origin:
Viet Nam.
Products Details:
Amount use
Each capsule, once per day.
Contraindications:
Hypersensitivity to Tenofovir Alafenamide or any of the excipients
Careful and informed
- Patients with decompensated liver failure. - Severe hepatitis. - CKD. - Renal toxicity. - Patients co-infected with HBV and hepatitis C or D. - Co-infection with hepatitis B virus and HIV. - Use in combination with other drugs. - Lactose intolerance.
See details